You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Health Update: First Convid-19 Vaccine tested in US shows positive immune response (Trump involvement in any part of this process is problematic)
- Thread starter playahaitian
- Start date
"First COVID-19 vaccine tested in the US shows promising immune response ahead of large trial" https://twitter.com/i/events/1283184709598457857
We are saved.
Damn thats cynical as f*ck
you mean you yourself are saved.We are saved.
Doesn't it take 5-10yrs of testing before they even start talking to the public about a vaccine??
Apparently this is historical because this is one of the first times damn near every country has decided to work TOGETHER on this ONE project...
Sharing information and resources at an unprecedented level.
So instead of being cynical about the result?
I'm sad as f*ck that the world is capable of doing something THIS super heroic and yet fails to do the same on other horrors plaguing us.
Last edited:
Aint nobody got time for datDoesn't it take 5-10yrs of testing before they even start talking to the public about a vaccine??
Aint nobody got time for dat
What you mean?? It's in your best interest they test these medical drugs.. You wanna be the first to take a shot and in 4yrs they come out telling those who got the first rounds of shot to get to ur doc asap.. Because something not lining up or how they did those copper color people in the syphilis test?? If your that safe with the medical industry, that's on you!!
Apparently this is historical because this is one of the first time damn near every country has decided to work TOGETHER on this ONE project
Sharing information and resources at an unprecedented level.
So instead of being cynical about the result?
I'm sad as f*ck that the world is capable of doing something THIS super heroic and yet fail to do the same on other horrors plaguing us
I dig what ur saying..
I'm saying don't be so quick when it comes to this medical treatments or test medicines.. If you wanna be first to raise your hand or jump up and down saying me me me!! That your risk
Big Money taking big hits because of COVID....the world moves fast when Big Money says its time to move fastApparently this is historical because this is one of the first times damn near every country has decided to work TOGETHER on this ONE project...
Sharing information and resources at an unprecedented level.
So instead of being cynical about the result?
I'm sad as f*ck that the world is capable of doing something THIS super heroic and yet fails to do the same on other horrors plaguing us.
What's in the vaccine?
Imagine putting COVID back in your system like they do with the flu shot. What are the long term affects of what's in this formula. BIG money about to start getting pumped somewhere for research and trials. Lot's of lives will be at risk too possibly.What's in the vaccine?
Big Money taking big hits because of COVID....the world moves fast when Big Money says its time to move fast
/thread
I dig what ur saying..
I'm saying don't be so quick when it comes to this medical treatments or test medicines.. If you wanna be first to raise your hand or jump up and down saying me me me!! That your risk
I gotta learn to be this succinct
What's in the vaccine?
No one knows!! That's why you have to keep your guard up!! Then again roll the dice and take the medical risk.. That's why they pay those folks money for taking the chance..
Wonder what the charge will be per dose......and of course the drug company will want a drug you need every year
I'm saying under $500 per year.. And hell yeah every year.. So billy gates and dr fauchi own the patent on the virus and they own the treatment and cure patent!! They won't release the cure til after 20+ yrs!! But billy gates will hit that zillionaire number before they start hinting on the cure shot!!
Was this trial given to people that were asymptomatic or those showing symptoms?
Was it given to people that were hospitalized and intubated or on ventilators?
How long do the antibodies stay and or do the number of antibodies substantially reduced over time?
these are the questions i need answers to before i start jumping for joy.
The last thing we need is the orange idiot taking credit for a vaccine to boost his chances for re-election.
'
Was it given to people that were hospitalized and intubated or on ventilators?
How long do the antibodies stay and or do the number of antibodies substantially reduced over time?
these are the questions i need answers to before i start jumping for joy.
The last thing we need is the orange idiot taking credit for a vaccine to boost his chances for re-election.
'
Doesn't it take 5-10yrs of testing before they even start talking to the public about a vaccine??
No. They fast tracked it.
To be expected, anyone thinking this vaccine ain't gone be some bullshit is lying to themselves, but any other source of this info besides a blog post?Not so FAST
Zerohedge
ZeroHedge - On a long enough timeline, the survival rate for everyone drops to zerowww.zerohedge.com
I want all you alarmists house negros who believe all the hype about how deadly the coronavirus hoax is to be first in line to inject yourself with this bullshit
Yep!!..just because they got to phase 2 .....they release news like this to pump up their stock price
"UK's Oxford University COVID-19 vaccine shows positive results in first phase of human trials" https://twitter.com/i/events/1285208837591449605
I'm cool on any vaccine. I'll wait until they test on all the white folks first.
AstraZeneca pauses coronavirus vaccine trial after unexplained illness in volunteer | CNN
Drug giant AstraZeneca said Tuesday it had paused global trials of its coronavirus vaccine because of an unexplained illness in one of the volunteers.
AstraZeneca pauses coronavirus vaccine trial after unexplained illness in volunteer
By Maggie Fox, CNN
Updated 11:49 AM ET, Wed September 9, 2020
(CNN)Drug giant AstraZeneca said Tuesday it had paused global trials of its coronavirus vaccine because of an unexplained illness in one of the volunteers.
"As part of the ongoing randomized, controlled global trials of the Oxford coronavirus vaccine, our standard review process triggered a pause to vaccination to allow review of safety data," the company said in a statement sent to CNN.
Another federal official raises questions about Trump's vaccine timeline
National Institutes of Health director Dr. Francis Collins told a Senate hearing Wednesday that it's a standard precaution in vaccine trials meant to ensure experimental vaccines don't cause serious reactions among participants.
"To have a clinical hold, as has been placed on AstraZeneca, as of yesterday, because of a single serious adverse event, is not at all unprecedented," Collins said at a hearing of the Senate Heath, Education, Labor and Pensions committee.
Content by CNN Underscored
How to sell your old tech before it loses its value.
CNN Underscored partnered with Decluttr to create this content. When you make a purchase, CNN receives revenue.
The company is testing its vaccine, called the Oxford vaccine because it was developed with Britain's University of Oxford, in the United States as well as the UK, Latin America, Asia, Europe and Africa.
"This is a routine action which has to happen whenever there is a potentially unexplained illness in one of the trials, while it is investigated, ensuring we maintain the integrity of the trials," the statement added.
close dialog
"In large trials, illnesses will happen by chance but must be independently reviewed to check this carefully. We are working to expedite the review of the single event to minimize any potential impact on the trial timeline. We are committed to the safety of our participants and the highest standards of conduct in our trials."
Here's how Trump could bigfoot the FDA and get a vaccine out ahead of the science
Earlier Tuesday, AstraZeneca joined eight other companies in signing a pledge promising they would not seek premature government approval for any coronavirus vaccine. They promised they would wait until they had adequate data showing any potential vaccine worked safely.
"We, the undersigned biopharmaceutical companies, want to make clear our on-going commitment to developing and testing potential vaccines for COVID-19 in accordance with high ethical standards and sound scientific principles," the statement read. It was signed by the CEOs of AstraZeneca, BioNTech GlaxoSmithKline, Johnson & Johnson, Merck, Moderna, Novavax, Pfizer and Sanofi.
"We believe this pledge will help ensure public confidence in the rigorous scientific and regulatory process by which COVID-19 vaccines are evaluated and may ultimately be approved," they added.
A coronavirus vaccine by Election Day? Probably not. Here's why
The AstraZeneca vaccine is one of three coronavirus vaccines in late-stage, Phase 3 trials in the US. A Data and Safety Monitoring Board (DSMB) usually monitors trials for adverse events and can order a pause or halt a trial, but AstraZeneca did not say who had stopped the trial.
Common adverse events in vaccine trials include fever, headache, soreness at the injection site and muscle pain.
Regulators and companies alike have been working to ensure people trust in the vaccine authorization process. The US Food and Drug Administration must give either emergency use authorization or full approval to any vaccine before it can be distributed in the US.
"This ought to be reassuring to everybody listening," Collins said at the hearing.
President Trump has said repeatedly he thinks at least one of the vaccines could be ready before Election Day on November 3. Most groups working on the vaccine say that is extremely unlikely but Biontech CEO and co-founder Ugur Sahin told CNN earlier Tuesday he believes the vaccine his company is developing with Pfizer could be ready for regulatory approval by mid-October.
Free the FDA and the CDC from political pressure
Dr. Tony Fauci, director of the National Institute of Allergy and Infectious Diseases, has said it's technically possible that if a DSMB saw strong evidence a vaccine was working before a trial was completed, it could recommend a halt to the trial so more people could get the vaccine. But Fauci said this was unlikely to happen before December.
However, discussions about early authorizations and reports about White House pressure to speed along a vaccine have worried the public, as well as former FDA officials. Former FDA commissioner Dr. Robert Califf told CNN last week he was concerned even before Covid-19 that Trump would pressure the FDA to make decisions he wanted.
"One of the things that's had me on pins and needles the last four years is literally Trump could do this with any drug or device or vaccine anytime he wants to," said Califf, who was commissioner from January 2016 through January 2018.
Fauci and FDA Commissioner Dr. Stephen Hahn have said they would not cave into political interference.
People shouldn't worry about safety of coronavirus vaccine trials, NIH director says
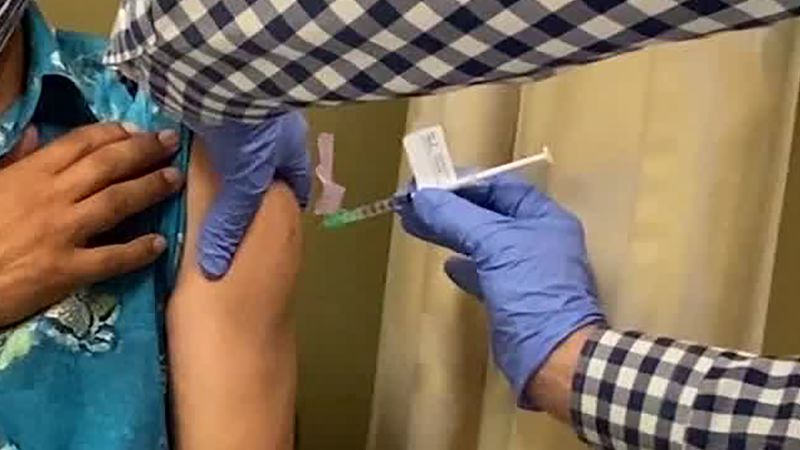
From CNN Health’s Maggie Fox
A participant receives a Covid-19 vaccination as she takes part in a vaccine study at Research Centers of America on August 7 in Hollywood, Florida. Joe Raedle/Getty Images
The various vaccines being tested to fight coronavirus are undergoing several layers of testing and people should not worry about their safety, National Institutes of Health Director Dr. Francis Collins said Thursday.
“We sped up this process in a variety of ways but not in a way to compromise safety. In fact, I would say these trials are more rigorous than any others that have ever been done for vaccines,” Collins told Dr. Sanjay Gupta and Anderson Cooper on CNN’s Global Town Hall.
“People ought to feel pretty comfortable. There are a lot of protective steps here that will keep this from running off the rails.”
The Data and Safety Monitoring Boards (DSMB), independent groups of mostly academic researchers who watch the data from the trials, are a firm line of defense keeping an eye out for any signal that an experimental vaccine is not working safely, Collins said.
From CNN Health’s Maggie Fox
A participant receives a Covid-19 vaccination as she takes part in a vaccine study at Research Centers of America on August 7 in Hollywood, Florida. Joe Raedle/Getty Images
The various vaccines being tested to fight coronavirus are undergoing several layers of testing and people should not worry about their safety, National Institutes of Health Director Dr. Francis Collins said Thursday.
“We sped up this process in a variety of ways but not in a way to compromise safety. In fact, I would say these trials are more rigorous than any others that have ever been done for vaccines,” Collins told Dr. Sanjay Gupta and Anderson Cooper on CNN’s Global Town Hall.
“People ought to feel pretty comfortable. There are a lot of protective steps here that will keep this from running off the rails.”
The Data and Safety Monitoring Boards (DSMB), independent groups of mostly academic researchers who watch the data from the trials, are a firm line of defense keeping an eye out for any signal that an experimental vaccine is not working safely, Collins said.
In fact, a DSMB is more likely to stop a trial if there’s evidence of a problem -- such as the illness in a volunteer that caused AstraZeneca to pause its vaccine trial this week, Collins said.“They are the only people, and they are not political people. They are the only ones who are watching to see who got the vaccine and who got the placebo and what happened,” he said.
“They aren’t going to raise their hand and say ‘hey its time to look at this, FDA,’ unless they see strong, statistically convincing data that this vaccine is working and it’s safe,” he added.
They are rushing this shit to try and capitalize on the market. I don't trust them at all.

